|
Schmidt T.G.M., Batz L., Bonet L., Carl U., Holzapfel G., Kiem K., Matulewicz K., Niermeier D., Schuchardt I. Overview of frequently used affinity tags and their corresponding affinity chromatography resin materials and elution methods References Low pH (~ pH 2-3) Synthetic c-Myc peptide 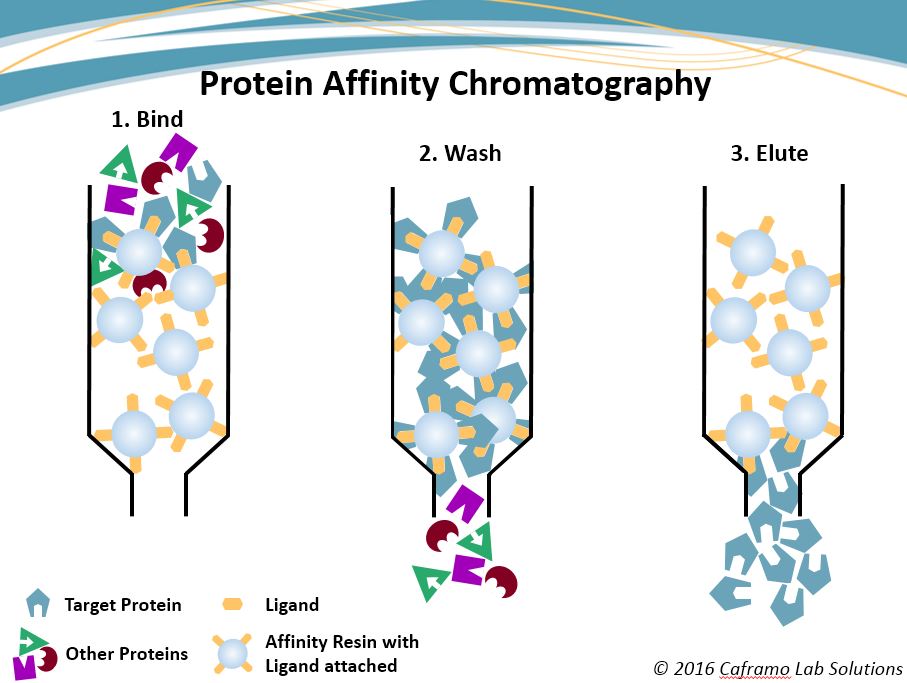
Immobilised transition metal ions: Ni 2+, Co 2+ (Ni-NTA, Talon)Ĭhitin-binding domain with self-splicing inteins The table below gives a brief overview of some commonly used affinity tags and their corresponding affinity chromatography resin materials and elution methods. The affinity chromatography resin you will use depends on the type of the affinity tag you have chosen during the construct design. Finally, the protein of interest is eluted from the affinity chromatography resin, usually either by adding a competitor for binding or a change in pH. 
Then a washing step is performed to remove weakly bound contaminants. You start with loading the cleared cell lysate (or cell culture medium in case of secreted proteins) onto the affinity chromatography resin to allow binding of your tagged protein. The goal of the affinity chromatography step is to separate your protein of interest from the majority of the host proteins.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
